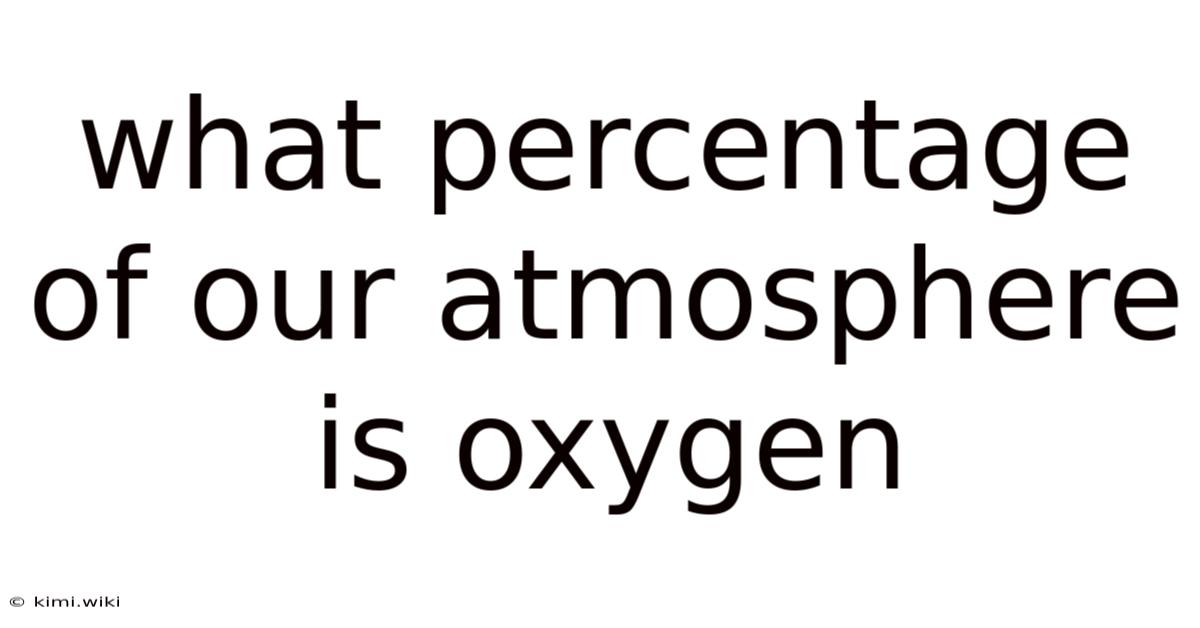What Percentage of Our Atmosphere is Oxygen? A Deep Dive into Earth's Vital Gas
Earth's atmosphere, that invisible blanket enveloping our planet, is a complex mixture of gases. While many components contribute to its overall composition, one gas stands out as crucial for life as we know it: oxygen. But just how much oxygen makes up our atmosphere? Understanding this percentage is vital to comprehending Earth's history, the evolution of life, and the delicate balance of our ecosystem. This article will walk through the precise percentage of oxygen in our atmosphere, explore its origins and importance, and address some frequently asked questions The details matter here..
Introduction: The Importance of Atmospheric Oxygen
The percentage of oxygen in Earth's atmosphere isn't a static number; it fluctuates slightly depending on location and altitude. And this seemingly small percentage is, in reality, a monumental figure, crucial for the survival of most life forms on our planet. On the flip side, a generally accepted average is around 20.Oxygen is the vital ingredient in aerobic respiration, the process by which most organisms convert energy from food. 95%. Without this readily available oxygen, life as we know it would be impossible.
This article will explore this crucial 20.95% in detail, examining its origins, the factors influencing its concentration, and its significance in shaping the Earth's environment and the evolution of life. We will also address common misconceptions and get into the scientific methods used to measure atmospheric oxygen levels.
The Composition of Earth's Atmosphere: More Than Just Oxygen
Before focusing solely on oxygen, it's crucial to understand the broader context of Earth's atmospheric composition. Our atmosphere is a dynamic system, a complex mixture of gases held in place by gravity. While oxygen holds a prominent position, it's far from the only component.
The major components of Earth's atmosphere, by volume, are:
-
Nitrogen (N₂): Approximately 78.08%. While inert in most biological processes, nitrogen is essential for life, playing a crucial role in the formation of amino acids and proteins.
-
Oxygen (O₂): Approximately 20.95%. This is the focus of our discussion, essential for respiration in most organisms.
-
Argon (Ar): Approximately 0.93%. A noble gas, argon is largely unreactive.
-
Carbon Dioxide (CO₂): Approximately 0.04%. Although a small percentage, carbon dioxide plays a vital role in the Earth's climate and is a key component of the carbon cycle. Its concentration is increasing due to human activities, leading to climate change And that's really what it comes down to..
-
Trace Gases: The remaining percentage includes other gases such as neon, helium, methane, krypton, hydrogen, and nitrous oxide. Although present in much smaller quantities, these gases can significantly impact atmospheric processes and the environment.
This composition is not uniform throughout the atmosphere. The concentration of gases varies with altitude, influenced by factors such as gravity, mixing processes, and chemical reactions.
The Origin of Atmospheric Oxygen: The Great Oxidation Event
The current level of atmospheric oxygen is a relatively recent phenomenon in Earth's history. Worth adding: early Earth's atmosphere was very different, lacking significant amounts of free oxygen. The primary source of oxygen in our atmosphere is photosynthesis, a process carried out by photosynthetic organisms, primarily plants and cyanobacteria (also known as blue-green algae).
The Great Oxidation Event (GOE), which occurred approximately 2.4 billion years ago, marked a turning point in Earth's history. Practically speaking, this period saw a dramatic increase in atmospheric oxygen levels, largely due to the evolution and proliferation of photosynthetic cyanobacteria. Before the GOE, oxygen was largely bound up in minerals and water. The release of free oxygen into the atmosphere had profound consequences, causing massive environmental changes and paving the way for the evolution of more complex life forms that depend on oxygen for respiration That's the part that actually makes a difference..
Factors Influencing Atmospheric Oxygen Levels: A Delicate Balance
The 20.95% oxygen concentration isn't simply a constant. Several factors influence its level, highlighting the delicate balance of Earth's ecosystems:
-
Photosynthesis: The primary source of oxygen, its rate is influenced by factors such as sunlight, temperature, water availability, and nutrient levels. Deforestation and other activities that reduce plant biomass can impact oxygen production.
-
Respiration: All aerobic organisms consume oxygen during respiration. The rate of respiration is influenced by the size and activity of the biosphere. Increased human population and industrial activity can lead to increased oxygen consumption That's the part that actually makes a difference. Nothing fancy..
-
Decomposition: Decomposition of organic matter consumes oxygen. Changes in land use and waste management practices can affect the rate of decomposition and thus oxygen levels Easy to understand, harder to ignore. Took long enough..
-
Oceanic Processes: The oceans play a crucial role in the oxygen cycle. Phytoplankton in the oceans produce a significant amount of oxygen through photosynthesis. Oceanic currents and water temperature also influence oxygen solubility and distribution.
-
Volcanic Activity: Volcanic eruptions release gases into the atmosphere, including some that can react with oxygen. While this impact is relatively small compared to other factors, it can influence local oxygen concentrations.
Measuring Atmospheric Oxygen: Scientific Methods
Accurately determining the percentage of oxygen in the atmosphere requires sophisticated scientific instruments. Some common methods include:
-
Gas Chromatography: This technique separates different gases in a mixture based on their physical and chemical properties, allowing for precise measurement of oxygen concentration Simple, but easy to overlook..
-
Paramagnetic Oxygen Analyzers: These instruments put to use the paramagnetic properties of oxygen to measure its concentration. Oxygen is attracted to a magnetic field, and this attraction is proportional to its concentration Not complicated — just consistent..
-
Electrochemical Sensors: These sensors measure oxygen concentration based on its electrochemical properties. These sensors are often used in portable oxygen analyzers The details matter here. But it adds up..
FAQ: Addressing Common Questions about Atmospheric Oxygen
Q: Is the percentage of oxygen in the atmosphere always constant?
A: No, the percentage fluctuates slightly due to natural processes and human activities. On the flip side, these fluctuations are generally small and within a relatively narrow range.
Q: Can the percentage of oxygen in the atmosphere ever become dangerously low or high?
A: Yes, significant changes in oxygen levels could have catastrophic consequences. A substantial decrease could be fatal to most life forms, while a significant increase could lead to increased fire risks and other environmental hazards Most people skip this — try not to..
Q: How does altitude affect the percentage of oxygen in the atmosphere?
A: The percentage of oxygen decreases with increasing altitude. This is because the atmosphere becomes less dense at higher altitudes, resulting in a lower concentration of all gases, including oxygen.
Q: What are the effects of decreasing oxygen levels in the atmosphere?
A: Decreasing oxygen levels can lead to various health problems in humans, including hypoxia (lack of oxygen to the body's tissues), reduced physical performance, and impaired cognitive function. In severe cases, it can be fatal. Decreasing oxygen levels can also affect other organisms in the ecosystem That's the part that actually makes a difference..
Conclusion: The Vital Role of Atmospheric Oxygen
The approximately 20.And understanding this balance is crucial for appreciating the importance of protecting our planet's ecosystems and mitigating the effects of human activities on atmospheric composition. Consider this: the delicate balance of our atmosphere, with its precious 20. But 95% of oxygen in Earth's atmosphere is not merely a statistic; it is a testament to the nuanced processes that have shaped our planet and enabled the evolution of life. This seemingly constant percentage represents a dynamic equilibrium, a delicate balance between oxygen production and consumption. Continued monitoring of atmospheric oxygen levels is essential for safeguarding the future of life on Earth. 95% oxygen, demands our respect, understanding, and diligent stewardship.