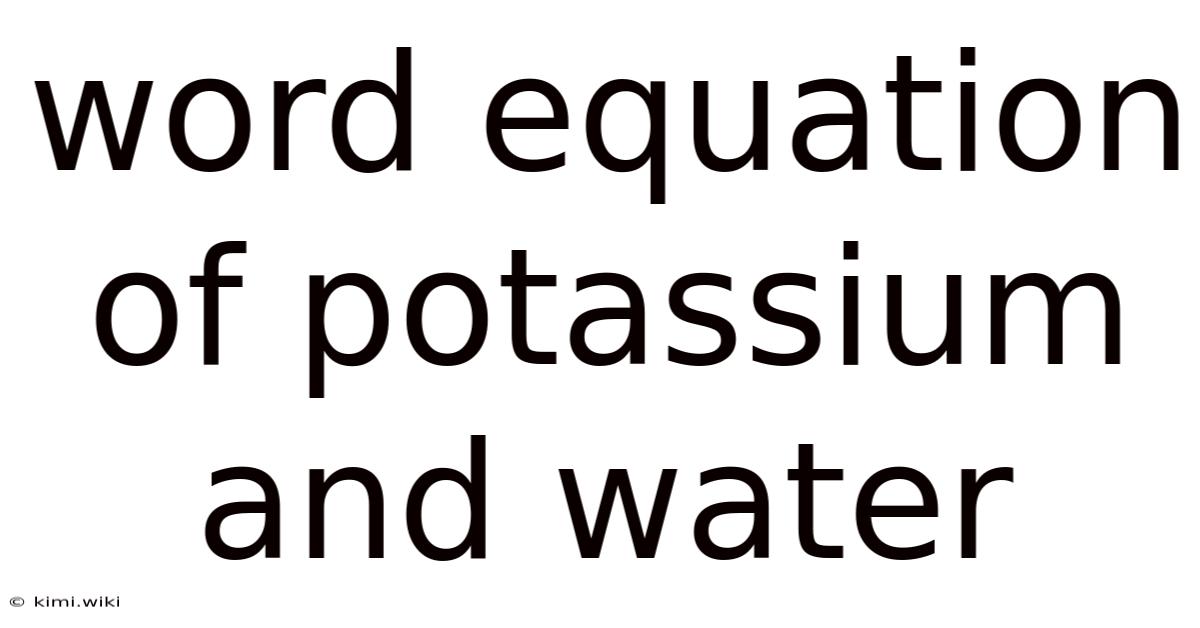The Reaction of Potassium and Water: A Deep Dive into the Word Equation and Beyond
The reaction between potassium and water is a dramatic and highly exothermic process, often used as a striking demonstration in chemistry classes. Understanding this reaction goes beyond simply memorizing a word equation; it involves grasping the underlying chemical principles, the safety precautions involved, and the implications of the reaction products. This article will provide a comprehensive exploration of the potassium and water reaction, covering everything from the basic word equation to the detailed details of the process Not complicated — just consistent..
Introduction: Understanding the Basics
The reaction between potassium (K) and water (H₂O) is a single displacement reaction, also known as a single replacement reaction. Worth adding: in this type of reaction, a more reactive element displaces a less reactive element from a compound. In this case, potassium, a highly reactive alkali metal, displaces hydrogen from water.
Potassium + Water → Potassium hydroxide + Hydrogen
While this provides a basic overview, it doesn't fully capture the energy released and the nuances of the reaction. Let's delve deeper into each aspect Simple, but easy to overlook..
The Reaction Mechanism: A Step-by-Step Breakdown
The reaction between potassium and water proceeds in several rapid steps:
-
Initial Contact: When potassium comes into contact with water, the highly reactive metal immediately begins to react with the water molecules. Potassium readily loses its single valence electron, becoming a positively charged potassium ion (K⁺) Still holds up..
-
Electron Transfer: This electron is transferred to a water molecule, reducing it. This reduction process often involves the formation of a hydroxide ion (OH⁻) and a hydrogen atom (H).
-
Hydrogen Formation: The hydrogen atoms produced then rapidly combine to form hydrogen gas (H₂). This is a highly exothermic step, releasing significant amounts of heat energy.
-
Potassium Hydroxide Formation: The potassium ions (K⁺) and hydroxide ions (OH⁻) combine to form potassium hydroxide (KOH), a strong alkali. Potassium hydroxide dissolves readily in water, resulting in a strongly alkaline solution.
The Equation in Detail: From Word Equation to Chemical Equation
The word equation, while helpful for beginners, lacks the precision of a chemical equation. The balanced chemical equation accurately reflects the stoichiometry of the reaction:
2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g)
This equation tells us:
- 2 moles of solid potassium (K(s)) react with 2 moles of liquid water (H₂O(l)).
- This produces 2 moles of aqueous potassium hydroxide (KOH(aq)) and 1 mole of hydrogen gas (H₂(g)).
The "(s)," "(l)," "(aq)," and "(g)" notations indicate the physical states of the substances: solid, liquid, aqueous (dissolved in water), and gas, respectively. This detailed representation is crucial for quantitative analysis and understanding the reaction's yield Simple, but easy to overlook..
The Energetics of the Reaction: Why is it so Exothermic?
The reaction is highly exothermic due to the significant difference in electronegativity between potassium and hydrogen. The transfer of the electron from potassium to water is a highly favorable process, releasing a substantial amount of energy in the form of heat. This heat often ignites the hydrogen gas, resulting in a characteristic flame. Still, potassium is an extremely electropositive element, meaning it readily loses its valence electron to achieve a stable electron configuration. Here's the thing — hydrogen, while not as electropositive as potassium, has a higher electronegativity than potassium. The energy released is enough to melt the potassium, which often appears as a silvery sphere moving rapidly across the water's surface.
Safety Precautions: Handling Potassium Safely
Potassium is a highly reactive alkali metal that reacts violently with water. Because of this, handling potassium requires extreme caution and adherence to safety protocols:
- Always wear appropriate personal protective equipment (PPE): This includes safety goggles, gloves resistant to alkali metals, and a lab coat.
- Perform the experiment in a well-ventilated area: The hydrogen gas produced is flammable and can be explosive in high concentrations.
- Use small quantities of potassium: Start with a very small piece (a few milligrams) of potassium to observe the reaction.
- Never perform the experiment unsupervised: Adult supervision is crucial, especially when conducting the experiment in educational settings.
- Have a fire extinguisher readily available: In case of uncontrolled burning or ignition, a fire extinguisher should be immediately accessible.
- Dispose of waste properly: The potassium hydroxide solution produced is highly alkaline and corrosive. Follow appropriate waste disposal procedures as outlined by your institution.
Observations and Demonstrations: What to Expect
When a small piece of potassium is added to water, several key observations can be made:
- Rapid reaction: The reaction occurs almost instantaneously upon contact.
- Movement of the potassium: The potassium melts into a silvery sphere due to the heat released, moving rapidly across the surface of the water.
- Hydrogen gas evolution: Bubbles of hydrogen gas are vigorously produced.
- Heat generation: Significant heat is released, evidenced by the boiling and steaming of the water.
- Flame: The hydrogen gas produced often ignites, resulting in a characteristic lilac-colored flame due to the excitation of potassium atoms.
- Alkaline solution: The resulting solution is strongly alkaline, turning red litmus paper blue.
Applications of the Potassium-Water Reaction:
While not a common industrial application, understanding this reaction is fundamental to several areas:
- Chemical education: The reaction serves as a powerful demonstration of the reactivity of alkali metals and the concept of exothermic reactions.
- Research in electrochemistry: The reaction's kinetics and energetics provide valuable insights into electrochemical processes.
- Understanding alkali metal behavior: Studying this reaction contributes to our wider understanding of the chemical properties of alkali metals and their interactions with other substances.
Frequently Asked Questions (FAQs)
-
Q: Can other alkali metals react with water similarly? A: Yes, other alkali metals like sodium, lithium, rubidium, and cesium also react with water, although the intensity of the reaction varies. Potassium's reaction is particularly vigorous.
-
Q: What makes potassium so reactive? A: Potassium's high reactivity stems from its electronic configuration. It has a single valence electron that is easily lost, resulting in a stable ion with a full electron shell. This ease of electron loss contributes to its high reactivity Easy to understand, harder to ignore. Surprisingly effective..
-
Q: Is the hydrogen gas produced pure? A: The hydrogen gas produced contains some water vapor due to the high temperature of the reaction.
-
Q: What are the potential hazards of this reaction? A: The main hazards are the highly exothermic nature of the reaction (potential burns), the production of flammable hydrogen gas (potential explosion), and the formation of a strongly alkaline solution (potential chemical burns) Simple, but easy to overlook..
-
Q: Can I perform this experiment at home? A: No. This experiment requires careful supervision and appropriate safety equipment. It should only be conducted in a controlled laboratory setting by trained personnel It's one of those things that adds up..
Conclusion: A Powerful Reaction with Broader Implications
The reaction between potassium and water is far more than a simple chemical equation; it's a powerful demonstration of fundamental chemical principles like reactivity, exothermic reactions, and redox processes. While its dramatic nature makes it a captivating experiment, it highlights the importance of safety precautions when handling reactive materials. In real terms, understanding this reaction deepens our appreciation of chemical reactivity and the energy transformations that occur in chemical processes. The insights gained from studying this reaction extend far beyond the simple word equation, illuminating critical concepts in chemistry and emphasizing the need for responsible scientific practice No workaround needed..